|
The F-C-H bond angle is 109.5 degrees in the tetrahedral CHF3 molecular geometry. It has a linear molecular geometry, given that there are only two atoms in this molecule. Both the atoms have lone pair of electrons and are sharing other electrons to complete their octets. Carbon and Oxygen atoms share a triple bond to fill their octets. Even though each of the bonds is polar, the molecule is non polar as the dipole moments cancel each other. Here is the Lewis structure for the molecule. Polar: IF, IF5, PC元 Nonpolar: CS2, SO3, SF6. Because the center atom, carbon, has three C-F and one C-H bonds with the three fluorine and one hydrogen atoms surrounding it. Carbon Monoxide or CO is a diatomic molecule. Explain how boiling points are determined and why they differ for each molecule. how many grams of cas is obtained if 152 g of cs2 is consumed in the reaction 11. 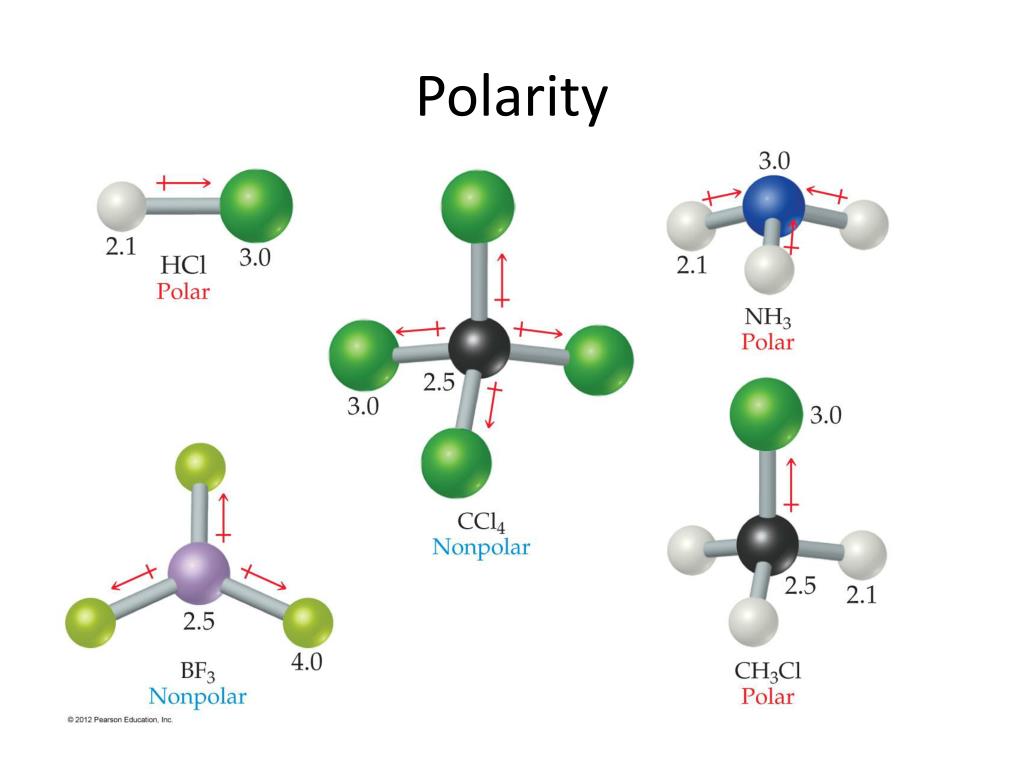
But bond polarity of C-S is canceled to each other in the linear. Determine the heat of evaporation of carbon disulfide, cs2(l) - cs2(g) given the enthalpies of reaction below. It has a difference in electronegativity values between sulfur and carbon atoms, with sulfur’s pull the electron cloud being slightly higher than carbon’s. Which one of these molecules has bonds that are most polar Rank the molecules in order of increasing polarity and explain why. The molecule of carbon disulfide (with linear shape CS2 molecular geometry) is tilted at 180 degree bond angle of S-C-S. 
Generally, solvents with dielectric constants greater than about 5 are considered "polar" and those with dielectric constants less than 5 are considered "non-polar. According to the VSEPR theory, the CHF3 molecule possesses tetrahedral molecular geometry. Consider the following molecules: BF3, SF6, BrF3, SCl2, CS2. However, as with many properties, the polarity is a continuous scale, and the correct question is not "is it polar or non-polar" but "how polar is it." Nonetheless, guidelines have been created to make it easier. Drag the appropriate items to their respective bins. Question: Identify whether each molecule given below is polar or nonpolar. Solvents are generally classified by the polarity, and considered either polar or non-polar, as indicated by the dielectric constant. This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
